Waves
Waves are defined as the movement of energy over a particular distance without the net movement of particles that mediate it. This basically means that when a wave is created energy is transferred from particle to particles causing them to move up and down (transverse) or side to side (longitudinal) but they always return to their original position.
The fundamental anatomy of a wave (amplitude, wavelength, frequency and speed) are all studied through year 8 – 10, so I will not spend time discussing them, instead, in this section, I will discuss, refraction, dispersion, interference and diffraction with references to famous experiments.
Refraction
Refraction is loosely defined as the bending of light through different mediums. So why does it happen? Well, when light is modelled as a wave and travels through different mediums, it interacts with the different particles within that medium. For example, when light travels through glass, the light interacts with the molecules of sodium carbonate, calcium carbonate and particles of sand, and these interactions slow down the passage of light through the lattice structure of glass. Comparatively, when light travels through air the particles that it interacts with are very loosely packed together as it is in a gaseous form, in fact, in the vacuum of space there are very, very few particles for the light to interact with hence it can follow a straighter path and thus travel at its true speed of 3*10^8 m/s. This holds true for other types of waves as well such as sound waves which also travel through a medium. So how does this relate to refraction? Well, as a beam of light travels between mediums of varying refractive indices, it bends towards or away from the normal. If the beam goes from high index to low index it bends away from the normal and vice versa.
Dispersion
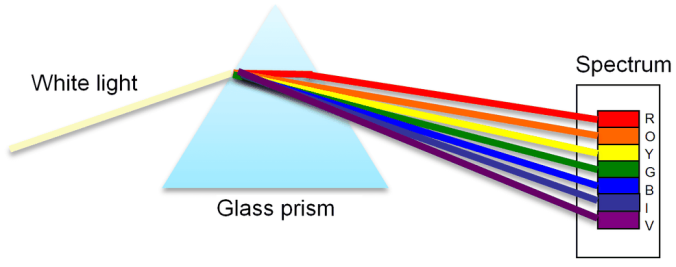
Dispersion is a similar concept to refraction in the sense that it involves the bending of light trough different materials. As we know, white light is a combination of the colours of the electromagnetic spectrum (ROYGBIV) and they all have slightly varying wavelengths and frequencies. When white light is shone through a glass prism, it splits into its component colours. This is because of their varying wavelengths, this is shown by the organisation of colours in a prism from the long wavelength at the top and low wavelength at the bottom as shown below.
Interference
Interference can be in 2 forms, constructive and destructive. Constructive interference is when the crests of one wave meet the crests of another wave. This causes the amplitude of the resultant wave to increase but retains the wavelength and the frequency. Destructive interference, on the other hand, is when the crest of a wave meets the trough of another wave, causing the resultant wave to have a decreased amplitude and maintain the wavelength and frequency. In 2 incident light waves, when their crests meet, they produce of very bright band of light.
Diffraction
Diffraction of waves is the deviation of waves around corners or small gaps. When a wave moves through a small gap, it bends away from the corners according to Huygens’s principle. It states that wavefronts (crests in 2-dimensions) are created by the interference of points emitting waves and when these points encounter a gap, the wave points on either end of the gap extend their wave out without restriction thus allowing diffraction to occur.
Wave-Particle Duality: The Debate of the Ages
For millennia physicists have debated over the nature of light, the key models put forward are the wave model and the particle model. The wave model suggests that light behaves like a wave and is a net transfer of light energy whereas the particle model suggests that light is a stream of particles like a beam of hot steam. 2 key experiments were conducted to determine the nature of light and are outlined below.
Young’s Double Slit Experiment
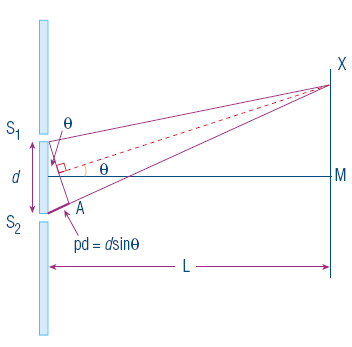
A famous experiment used to determine the wave nature of light was Young’s double slit experiment. Although the original intention of the experiment had to do with optics rather than quantum mechanics it still supports the wave model of light elegantly. The experiment is set up with a detection screen placed a known distance away from a thick opaque film that has 2 very small slits in it. Monochromatic light is shone through these slits and the pattern is observed on the screen. If the particle model of light is to be accepted, then there should be 2 clear bands of light however the result of the experiment showed a series of alternating bright and dark bands. This was a monumental discovery as it suggested that not only did light diffract but also interfere. The dark bands of the pattern indicate destructive interference where the crests of light from one slit meet the trough of the light from the second slit. This is caused by the length of distance that 2 beams of light that have to travel, this difference is known as the path difference. This shows that as light exhibits phenomenon which is exclusive to waves, it must be a wave. The experiment is outlined below.
The Photoelectric Effect
The photoelectric effect is an experiment which contradicts the wave model of light. A beam of monochromatic light is shone on a particular metal and this results in a release of electrons from the lining of the metal. These electrons are detected by a wire and a current is observed by the sensor. This is known as the photo-current. This is fairly simple stuff, energy is applied to a metal and the electrons get excited and they leave the surface of the metal. A contradiction occurs however when it is found that there is essentially a 0-time delay between the light switched on and the detection of a photo-current, which is inconsistent with the wave model, which suggests that there should be a time delay to allow for the energy to be fully absorbed. Another contradiction is that upon increasing the intensity of light the total energy of the electrons was constant (ie kinetic as measured using the stopping voltage, refer to Units 4 Physics textbook) but the photo-current was increased. And finally, there was a certain amount of frequency required to free the electrons, any frequency below that wouldn’t work, not even an increase in intensity.
So… A dilemma
This was a problem, there was good evidence for and against the wave model but which one was correct? Well, scientists theorised that maybe light was a stream of particles, called photons, that would hit and be absorbed by the electrons on the metal sheet in the photoelectric experiment. This would explain why there was an instantaneous release of electrons and why increasing the intensity (ie increasing the number of photons) would result in more electrons being released (due to more collisions). But this still didn’t fully explain how a particle like a photon could diffract and interfere. Well, it was theorised that maybe it was both, when we looked for wave properties we could find them and when we looked for particle properties we could find them too. This led to the development of the Theory of Wave-Particle Duality.
Wow… this was really long, next week I will finish off Wave-Particle Duality and start talking about Heisenberg’s cat and some Quantum Tunneling, so tune in and tell your friends to read this series!!

 In this 11-part, fortnightly series we will venture through the realms of particle physics, quantum mechanics and superstring theory to devise a set of laws to describe the universe so join me, Nipun Kapadia, on this journey to determine the Laws of the Universe.
In this 11-part, fortnightly series we will venture through the realms of particle physics, quantum mechanics and superstring theory to devise a set of laws to describe the universe so join me, Nipun Kapadia, on this journey to determine the Laws of the Universe.